Last Updated: March 2026 | Reading Time: 9 minutes | ~2,000 words
India is the world’s largest consumer of antibiotics — and simultaneously the country with one of the highest burdens of antibiotic-resistant infections globally. The WHO has declared Antimicrobial Resistance (AMR) one of the top 10 global public health threats; in India, AMR is estimated to cause over 300,000 deaths annually — making it deadlier than malaria, HIV, and road traffic accidents combined. The drivers are well-documented and uniquely powerful in India: over-the-counter antibiotic sales without prescription (legal only with prescription but widely available); self-medication with “leftover” antibiotics; physician over-prescribing driven by diagnostic uncertainty, patient pressure, and perverse commercial incentives; agricultural antibiotic use contaminating water and food; inadequate laboratory diagnostic infrastructure meaning antibiotics are given before knowing whether infection is bacterial or viral. The tragedy: infections that were once curable with penicillin now require last-resort antibiotics; infections once treated in one day of IV therapy now require 14-day ICU courses; and surgical procedures that were once routine are becoming dangerous in the absence of effective prophylactic antibiotics.
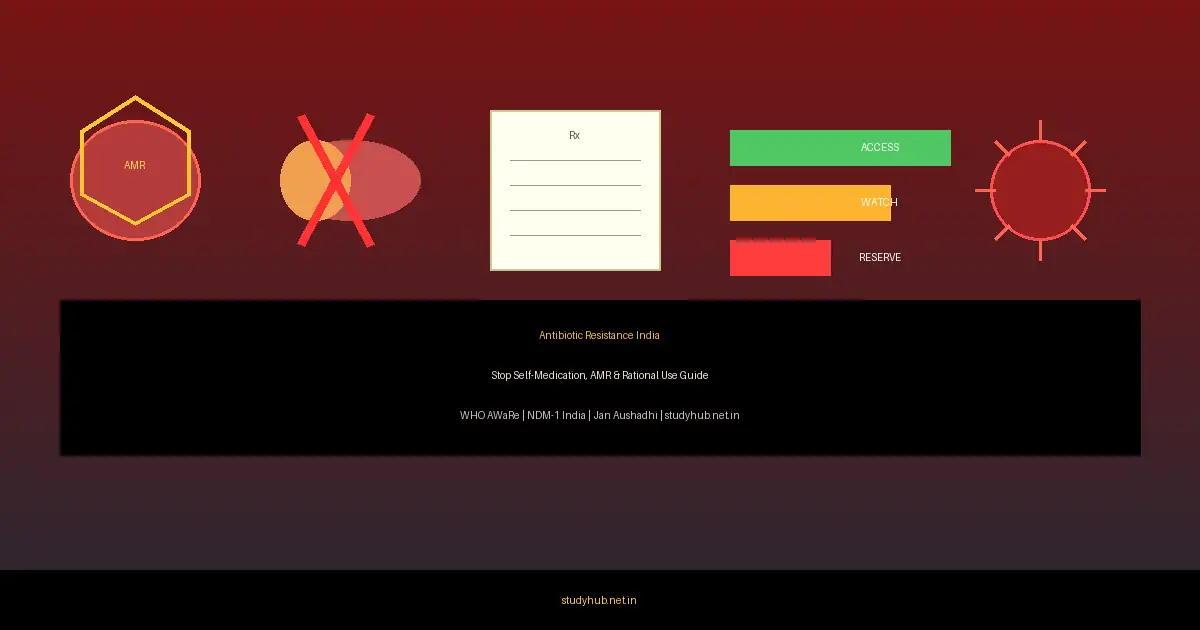
The WHO AWaRe Classification — Understanding Antibiotic Tiers
| AWaRe Category | Examples | When Used | India Context |
|---|---|---|---|
| ACCESS — First-line, essential (target: >60% of antibiotic use) | Amoxicillin; phenoxymethylpenicillin (penicillin V); doxycycline; trimethoprim; nitrofurantoin; metronidazole; cloxacillin; ampicillin; gentamicin; azithromycin; co-amoxiclav | Community-acquired pneumonia; simple UTI; skin infections; dental infections; uncomplicated Shigella; Lyme disease | India’s Access antibiotic use ratio is approximately 50% — below WHO target; amoxicillin and azithromycin widely available without prescription; co-amoxiclav often prescribed instead of amoxicillin unnecessarily → Watch-to-Access ratio imbalanced |
| WATCH — Higher resistance risk; use only when first-line fails or specific indication (should be ≤40%) | Ciprofloxacin; levofloxacin; cephalosporins (cefixime, ceftriaxone, cefpodoxime); azithromycin for typhoid; clarithromycin; piperacillin-tazobactam; vancomycin; meropenem; ertapenem; linezolid | Complex UTI; CAP failing amoxicillin; typhoid (fluoroquinolone or cephalosporin); hospital-acquired infections; MRSA (vancomycin); Pseudomonas (piperacillin) | Fluoroquinolones (ciprofloxacin, levofloxacin) grossly overused in India — prescribed for viral URTI, mild gastroenteritis, undifferentiated fever; ciprofloxacin resistance in E. coli UTI in India now >70% in some surveys (rendering empirical UTI treatment ineffective); cephalosporin use growing without culture guidance |
| RESERVE — Last resort; use only when all others fail (should be <2%) | Colistin; polymyxin B; ceftazidime-avibactam; cefiderocol; tigecycline; fosfomycin IV; temocillin; new β-lactam/β-lactamase inhibitor combinations | Carbapenem-resistant organism (CRO) infections — KPC, NDM, OXA-48; pan-drug resistant Klebsiella, Acinetobacter; CRKP (carbapenem-resistant Klebsiella pneumoniae) | India identified the New Delhi Metallo-β-lactamase (NDM-1) enzyme in 2010 — a globally spreading resistance mechanism named after India; colistin resistance rising in Indian ICUs; Reserve antibiotics available only at major hospitals; treatment failure from these organisms carries 40–60% mortality |
India’s AMR Crisis — Data & Drivers
| Driver | India-Specific Data | Solution |
|---|---|---|
| OTC antibiotic sales without prescription | 68–75% of antibiotic purchases in India are without a doctor’s prescription (CDDEP 2021 survey); pharmacists sell antibiotics routinely based on self-reported symptoms; antibiotics are among India’s top 5 OTC purchases; New Delhi Study: 77% of antibiotic courses purchased were for viral illnesses (for which antibiotics are useless) | Strict enforcement of prescription-only antibiotic sales; Schedule H and H1 antibiotic regulations require prescriptions but enforcement is weak; digital prescriptions linked to pharmacy dispensing; public campaigns on harm of OTC antibiotic use |
| Incomplete antibiotic courses | Cultural practice: stop antibiotics when symptoms improve (typically 2–3 days), leaving sub-therapeutic levels that kill susceptible bacteria but allow resistant mutants to survive and multiply; widespread belief that antibiotics are “hot” medicines that should not be taken for long course | Clear patient education at the point of prescription: always complete the full course; explain why 3-day stopping is more dangerous than completing 5–7 days; prescribe exact number of tablets (not “1 strip” — which may have more or fewer than needed) |
| Antibiotic prescribing for viral illness | Studies show 40–60% of antibiotic prescriptions in Indian primary care are for URTI (common cold, flu, viral sore throat) — conditions caused by viruses where antibiotics have zero benefit; URTI causes: rhinovirus, influenza, RSV — all viral; antibiotics do not reduce duration, severity, or complications of viral URTI; they do cause: antibiotic-associated diarrhoea, microbiome disruption, selection of resistant organisms, allergic reactions, cost | Implement rapid diagnostic tests (strep antigen test for pharyngitis — only Group A strep warrants antibiotic; everything else is viral); physician education on delayed prescribing strategy; patient education campaigns (“Antibiotics are not for colds”) |
| Agricultural antibiotic use | India uses approximately 3,000 tonnes of antibiotics annually in livestock and aquaculture (CDDEP estimates); colistin was used extensively in poultry until 2019 ban — mcr-1 gene (colistin resistance) found in Indian poultry and human gut bacteria; tetracyclines used in fish farming contaminate water bodies; antibiotic-resistant bacteria and resistance genes spread from farm-to-fork and via water | Enforce colistin ban; restrict agricultural antibiotic use to veterinary prescription; invest in alternative disease prevention in livestock (vaccines, biosecurity, probiotics); monitor resistance in environmental samples |
Frequently Asked Questions
When do I actually need an antibiotic?
This is the most practically important question for every Indian patient and family — because the reflexive “give antibiotic” response to fever, sore throat, or cough is the primary driver of AMR and exposes patients to risk without benefit: CONDITIONS THAT DO NOT NEED ANTIBIOTICS (viral causes — antibiotics have zero effect): Common cold (rhinovirus, coronavirus, adenovirus): runny nose + mild sore throat + low fever; antibiotics do nothing; treat with paracetamol for fever/aching, nasal saline drops, rest, fluids, honey for cough (1 year+). Influenza (flu — influenza A/B virus): fever + body aches + dry cough + headache + fatigue; antivirals (oseltamivir) may help if given within 48h of symptom onset in high-risk patients; antibiotics are useless and harmful. Viral gastroenteritis (stomach flu — norovirus, rotavirus): nausea + vomiting + diarrhoea without blood; ORS + zinc + continue feeding; no antibiotic. Viral pharyngitis (most sore throats are viral — adenovirus, EBV): antibiotics not warranted unless rapid strep test positive for Group A Streptococcus. Dengue, chikungunya: viral — antibiotics counterproductive; the fever IS NOT bacterial. CONDITIONS THAT USUALLY DO NEED ANTIBIOTICS: Bacterial pneumonia (fast breathing + fever + consolidation on X-ray): amoxicillin first-line. Urinary tract infection (burning urination + frequency without URTI symptoms): nitrofurantoin or trimethoprim first-line for uncomplicated UTI women (culture-guided). Streptococcal pharyngitis (Group A Strep confirmed): amoxicillin × 10 days (prevents rheumatic fever — very important in Indian children). Typhoid: azithromycin (uncomplicated) or ceftriaxone (hospitalised, severe). Bacterial diarrhoea — bloody (dysentery — Shigella): azithromycin 3 days. Dental abscess: amoxicillin or clindamycin. Skin and soft tissue infection — cellulitis: amoxicillin-clavulanate or cloxacillin. The practical message: If you have fever + runny nose + mild sore throat → you almost certainly have a viral URTI → you do NOT need an antibiotic. If your physician prescribes one → ask “Is this infection definitely bacterial? What specific bacteria are we treating? What is the evidence this antibiotic helps here?” These are reasonable questions that protect you from unnecessary antibiotic exposure.
What is antibiotic resistance and why does it matter to me personally?
Antibiotic resistance is often presented as a distant, abstract global threat — but for Indian patients, it is a present, personal, and potentially lethal risk: How resistance develops — explained simply: Any antibiotic course kills most bacteria — but in any population of billions of bacteria, some have random genetic mutations making them slightly less susceptible to the antibiotic. These survive, reproduce, and pass on their resistance genes — to their offspring AND to other bacteria species via horizontal gene transfer (plasmids). This process is continuous, unavoidable, and accelerates with every unnecessary antibiotic course. The more antibiotics used inappropriately, the faster resistance develops. How it affects YOU specifically: Imagine you have a kidney infection (pyelonephritis) — E. coli is the most common cause. Historically, ciprofloxacin would cure it in 5–7 days. But ciprofloxacin resistance in E. coli in India is now >70% in many hospitals — meaning 70% chance your infection is already resistant to the antibiotic you’d normally get. You need a culture test, wait 48 hours for results, and may need IV antibiotics for what was once a 5-day oral tablet course. Or: you have a surgical procedure in an Indian hospital. Prophylactic antibiotics are given to prevent wound infection. If your gut bacteria (carried into the surgical field) are resistant to the prophylactic antibiotic → higher risk of surgical site infection, sepsis, death — for a routine operation. The “AMR is someone else’s problem” fallacy: Every unnecessary antibiotic course you take increases resistance in YOUR gut bacteria — creating a personal reservoir of resistance genes that will affect YOUR future infections. India’s AMR burden is your personal AMR burden. India’s specific resistance crisis data: Carbapenem-resistant Klebsiella pneumoniae: 12–15% of ICU infections in Indian tertiary hospitals are carbapenem-resistant — virtually untreatable. Methicillin-resistant Staphylococcus aureus (MRSA) hospital-acquired infections: 40–50% of S. aureus in Indian ICUs → requires vancomycin, not standard antibiotics. Multidrug-resistant tuberculosis (MDR-TB): 130,000 new cases annually in India — the global epicentre of drug-resistant TB. NDM-1 (New Delhi Metallo-β-Lactamase): discovered in India 2010, now spread to 100+ countries — named after India to the nation’s significant embarrassment and public health detriment.
Are generic medicines the same as branded antibiotics?
Generic vs branded antibiotic confusion is one of the most consequential pharmacological misunderstandings among Indian patients — leading to unnecessary spending on branded medicines when generics are therapeutically identical: The regulatory framework: A generic medicine contains the same active ingredient in the same dose, dosage form (tablet/capsule/injection), and route of administration as the branded original. Under Indian drug regulation (CDSCO), generic medicines must demonstrate bioequivalence — same absorption, distribution, and blood concentration profile — to the branded originator. Therefore: Amoxicillin 500mg manufactured by Cipla (Mox) is therapeutically identical to Amoxicillin 500mg by GlaxoSmithKline (Amoxil) — same molecule, same dose, same effect. Cost difference — enormous in India: Branded amoxicillin 500mg × 10 caps: ₹120–180. Generic amoxicillin 500mg × 10 caps (Jan Aushadhi): ₹8–20. The difference (up to 10×) represents no therapeutic difference — only marketing costs and brand premium. PM Jan Aushadhi Kendras (now 10,000+ stores across India, 2024): provide generic equivalents of 1,800+ medicines including commonly prescribed antibiotics at dramatically lower cost; quality-assured through government procurement. Where to find generics: Jan Aushadhi Kendras (government generic stores — blue signage); ask your doctor specifically for the INN (International Non-proprietary Name) — the generic name — rather than the brand name; PMBJP (Pradhan Mantri Bhartiya Janaushadhi Pariyojana) medicines are government quality-assured. The important caveats: Not all generic manufacturers have equivalent quality control — choosing government-procured or established manufacturer generics (Cipla, Sun Pharma, Dr Reddy’s generic ranges) is prudent; for narrow therapeutic index drugs (not typically antibiotics — more relevant for phenytoin, warfarin, digoxin, thyroid drugs), bioequivalence is more critical; for standard antibiotics (amoxicillin, doxycycline, azithromycin, ciprofloxacin), generic equivalents from reputable manufacturers are clinically interchangeable. The most rational choice for most antibiotic courses: Jan Aushadhi or established Indian generic manufacturer at fraction of branded cost.
What are India’s national AMR action plan and regulations?
India has developed significant policy infrastructure to combat AMR — the implementation gap remains the critical challenge: National Action Plan on AMR (NAP-AMR) 2017–2021 (India): India’s first comprehensive AMR strategy, aligned with the WHO Global Action Plan. Five strategic objectives: Awareness and education; Surveillance (ICMR-AMR network of 20+ sentinel surveillance sites collects standardised resistance data from across India); Infection prevention and control; Antibiotic use optimisation; Research and innovation. Key regulatory measures: Schedule H1 (2013): Added 24 critical antibiotics (including fluoroquinolones, carbapenems, cephalosporins, vancomycin) to Schedule H1 — requiring pharmacy-written record of prescription; pharmacist must record prescriber details; package must carry “Caution: To be sold by retail on the prescription of a Registered Medical Practitioner only” red label. The implementation gap: Schedule H1 enforcement is minimal; only large organised pharmacies in metro cities consistently require prescriptions; neighbourhood retail pharmacies routinely sell H1 antibiotics without prescriptions. Hospital Infection Control Committees (HICCs/ICCs): Required at all hospitals under Standards for Safety and Standards accreditation; responsible for antibiotic stewardship programmes (ASPs), formulary management, and surveillance. Antibiotic Stewardship Programmes (ASPs): ICMR and NABH mandate ASPs at tertiary hospitals — include restriction policies (requiring ID specialist approval for Reserve antibiotics), de-escalation protocols (switching IV to oral, broad to narrow spectrum once cultures available), and 72-hour antibiotic review. The “Red Line” campaign: WHO-endorsed awareness campaign; medicines with a red line on packaging indicate prescription-only status; community campaigns in India to educate on prescription requirement; limited penetration in rural areas. Jan Vishwas Act 2023: Decriminalised minor offences including Schedule E (but NOT Schedule H/H1) violations — controversy about whether this reduces deterrence for non-prescription antibiotic selling; government clarified Schedule H remains criminal violation; ongoing advocacy needed. What you can do: Observe the red line: Never buy antibiotics without a doctor’s prescription. Complete the course: Never stop antibiotics midway or “save” leftover antibiotics. Dispose safely: Return unused antibiotics to pharmacies or authorised disposal — do not flush down drain (environmental resistance reservoir). Wash hands regularly: Prevents transmission of resistant bacteria in hospitals and community. Advocate: Support mandatory antibiotic stewardship policies at hospitals you attend.
What is antibiotic prophylaxis and when is it appropriate?
Antibiotic prophylaxis — giving antibiotics before an event to prevent infection, rather than to treat an existing infection — is one of the most commonly misused antibiotic practices in India, contributing significantly to AMR: APPROPRIATE prophylaxis situations (evidence-based): Surgical prophylaxis: A single dose of antibiotic given within 60 minutes before skin incision significantly reduces surgical site infection (SSI) — cefazolin is the gold standard for most clean-contaminated surgeries; the dose is stopped at 24 hours (no benefit to extending beyond 24 hours post-surgery, yet many Indian surgeons prescribe oral antibiotics for 5–7 days post-operatively — this is AMR-harmful and evidence-free). Rheumatic fever secondary prophylaxis: Monthly benzathine penicillin G IM for children with previous rheumatic fever/rheumatic heart disease — prevents streptococcal throat infection from re-triggering cardiac inflammation; recommended for 5 years minimum or until age 21. Dental prophylaxis for infective endocarditis: Amoxicillin single dose before invasive dental procedures in patients with high-risk cardiac conditions (prosthetic heart valves, complex congenital heart disease, previous endocarditis). Malaria prophylaxis for travel to endemic areas: Doxycycline, mefloquine, or atovaquone-proguanil — appropriate and proven. UTI prophylaxis in recurrent UTI women: Low-dose nitrofurantoin or trimethoprim nightly — evidence-based for women with ≥3 UTIs/year. INAPPROPRIATE prophylaxis (widespread in India — contributing to AMR): “Post-operative course” for 5–7 days after clean surgery: No evidence of additional benefit beyond a single pre-operative dose; pure AMR contribution. Antibiotics for viral URTI “to prevent secondary bacterial infection”: Zero RCT evidence supporting this practice; major driver of antibiotic misuse in Indian private practice. Antibiotics before every dental procedure regardless of cardiac risk: Appropriate only for the specific high-risk cardiac conditions listed by AHA/ESC guidelines — overused in India for all patients. “Seasonal” antibiotic courses during viral flu season: Self-prescribed or prescribed by chemists — completely irrational; viral infections require no antibiotics; antibiotics do not prevent viral infections from progressing to bacterial complications. The stewardship principle: Every antibiotic course must have three things justified: the indication (what infection/prevention); the agent (why this antibiotic rather than a narrower-spectrum option); the duration (why this length). “When in doubt, leave it out” is the foundation of rational antibiotic use.
What to Read Next
- Tuberculosis — MDR-TB India: 130,000 Cases/Year — Caused by Incomplete TB Treatment
- Typhoid — Antibiotic-Resistant Typhoid (XDR-Typhoid) Emerging in India
- Diarrhoea — Most Childhood Diarrhoea in India Does NOT Need Antibiotics
- Dengue — Antibiotics for Dengue Are Harmful and Useless; Dengue is Viral
- Pneumonia — Amoxicillin: The Right Antibiotic for Pneumonia; Don’t Use Cephalosporins Unnecessarily
In 1928, Alexander Fleming discovered penicillin. In his Nobel Prize acceptance speech in 1945, he warned: “It is not difficult to make microbes resistant to penicillin in the laboratory by exposing them to concentrations not sufficient to kill them… The time may come when penicillin can be bought by anyone in the shops. Then there is the danger that the ignorant man may easily underdose himself and by exposing his microbes to non-lethal quantities of the drug make them resistant.” He was describing India’s pharmacies in 2026 with uncanny precision. The warning is 80 years old. The crisis is now.
About This Guide: Written by the StudyHub Health Editorial Team (studyhub.net.in) based on WHO Global Action Plan on AMR, ICMR National AMR Surveillance Network, and India NAP-AMR guidelines. Last updated: March 2026.
Authoritative Sources: WHO AMR Fact Sheet | ICMR AMR India | CDDEP — Antibiotic Use India
🚫 Never do this: Buy antibiotics without a doctor’s prescription. Stop the course after 2–3 days when you feel better. Give your antibiotic leftovers to family members. Take antibiotics for a cold/flu. These actions directly create antibiotic resistance that puts your own future health at risk.
⚕️ Medical Disclaimer: This article provides general information about antibiotic use and AMR. All antibiotic treatment decisions must be made by a qualified healthcare provider based on individual clinical assessment. Never self-prescribe, self-diagnose, or self-discontinue antibiotics without medical guidance.
