Last Updated: March 2026 | Reading Time: 9 minutes | ~2,000 words
Thalassaemia is a group of inherited autosomal recessive haemoglobin disorders caused by reduced or absent synthesis of one or more globin chain subunits of haemoglobin. India carries the world’s largest burden of thalassaemia: approximately 40–45 million beta-thalassaemia trait carriers (HbA/HbA2-elevated — silent carriers) — approximately 3–4% of the general Indian population — with carrier rates rising to 8–17% in specific communities (Sindhis, Punjabis, Gujarati Lohanas, Bengali Muslims, Kodavas of Coorg). Approximately 10,000–15,000 thalassaemia major (beta-thalassaemia major — BTM) children are born in India annually, joining the existing pool of approximately 1–1.5 lakh (100,000–150,000) thalassaemia major patients — who require regular blood transfusions every 2–4 weeks for life. Without access to regular transfusions and iron chelation therapy, thalassaemia major children die in childhood from severe anaemia and cardiac failure from iron overload. India’s thalassaemia burden represents an enormous, expensive, and preventable public health crisis — expensive because each child with thalassaemia major requires approximately ₹5–8 lakh per year in treatment costs (transfusions + chelation + monitoring) lifelong; preventable because universal premarital and antenatal carrier screening can identify at-risk couples and prevent virtually all new BTM births through prenatal diagnosis. India’s National Thalassaemia Control Programme and emerging gene therapy trials (LentiGlobin, CRISPR-Cas9 based — Casgevy) offer hope for the future.
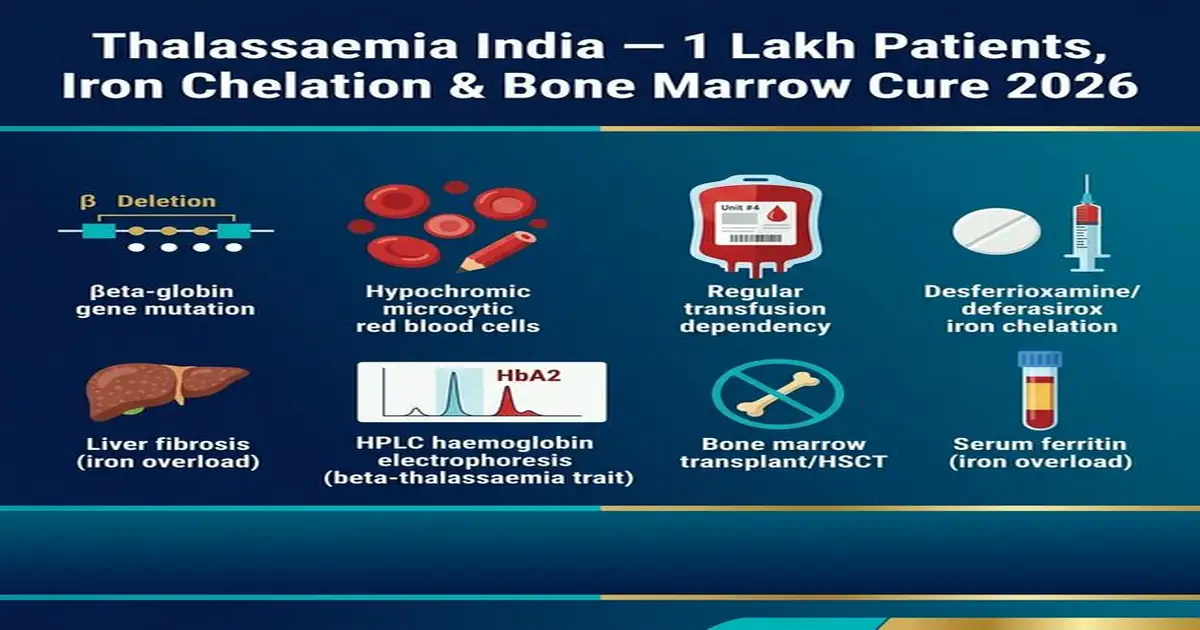
Thalassaemia India — Types, Transfusion, Iron Chelation and Cure
| Category | Description / Mechanism | Management | India Context |
|---|---|---|---|
| Beta-Thalassaemia Major (BTM) — Transfusion-Dependent | Homozygous or compound heterozygous beta-globin gene mutations → severely reduced/absent HbA production; severe haemolytic anaemia (Hb 2–5 g/dL untreated); massive erythroid hyperplasia (bone marrow expands to compensate → “chipmunk facies” — frontal bossing, maxillary prominence; skull X-ray “hair-on-end” appearance — cortical thinning + trabecular expansion — pathognomonic); extramedullary haematopoiesis (liver, spleen, paravertebral masses — mediastinal masses on CXR); hepatosplenomegaly; growth retardation; endocrine failure (hypogonadism, diabetes — from iron overload); cardiac failure (iron-overload cardiomyopathy — leading cause of death in BTM without chelation); diagnosis: HPLC — HbA absent or markedly reduced; HbF very high (60–98%); HbA2 variable; CBC: severe microcytic hypochromic anaemia + target cells + nucleated RBCs on peripheral smear | Regular blood transfusion: every 2–4 weeks; target pre-transfusion Hb 9–10 g/dL; leukodepleted, CMV-negative, blood group phenotype-matched (reduce alloimmunisation — C, E, Kell antigens); each unit transfused adds ~225 mg iron → cumulative iron overload; hepatic iron overload (liver cirrhosis); cardiac iron overload (cardiomyopathy — T2* MRI cardiac iron — most important monitoring); endocrine iron overload (pancreas → diabetes; pituitary → delayed puberty; thyroid); Iron chelation therapy (ICT): MANDATORY — starts when serum ferritin >1000 µg/L or after 10–20 units transfused; Deferasirox (DFX — Exjade/Asunra — oral): 20–40 mg/kg/day — most widely used India (once daily oral tablet); Deferoxamine (DFO — Desferal — SC/IV): 25–50 mg/kg/day subcutaneous infusion overnight (8–12h) via portable infusion pump; cumbersome but gold standard; Deferiprone (DFP — Ferriprox — oral): 25 mg/kg TDS; particular benefit for cardiac iron chelation (crosses cardiac cell membranes better than DFX); combination chelation: DFP + DFO (“super-chelation”) for severe cardiac iron overload; monitoring: serum ferritin monthly; LIC (liver iron concentration) — T2* MRI liver (gold standard); T2* cardiac MRI (cardiac iron — critical — correlates with cardiac mortality) | India thalassaemia registry: approximately 1–1.5 lakh BTM patients; free transfusion: government blood banks — available in major cities; rural access very poor; iron chelation India: Deferasirox (generic — Deferasirox Cipla/Sun Pharma): ₹60–80/tablet; 30kg child on 30mg/kg/day needs 900mg/day (~1 tablet 500mg + ½) = ₹90–120/day = ₹3,000–4,000/month for chelation alone; DFO SC pump: ₹200–400/infusion × 5 nights/week = ₹4,000–6,000/month; total treatment cost (transfusion + chelation + monitoring): ₹5–8 lakh/year; PMJAY (Ayushman Bharat): thalassaemia transfusion and chelation covered for beneficiaries (though implementation inconsistent); Thalassaemia International Federation (TIF) — India chapter: advocacy for free chelation national programme |
| Beta-Thalassaemia Intermedia & Trait | Beta-Thalassaemia Intermedia (BTI — non-transfusion-dependent thalassaemia — NTDT): compound heterozygous with milder mutations; Hb 7–10 g/dL; does not require regular transfusions for survival but may need transfusions during stress (pregnancy, surgery, infection, growth spurts); complications: iron overload still occurs (dietary iron hyperabsorption due to ineffective erythropoiesis even without transfusion); extramedullary haematopoiesis masses; thromboembolic risk (hypercoagulable); splenomegaly; pulmonary hypertension; leg ulcers; Beta-Thalassaemia Trait (BTT — carrier — heterozygous): ONE mutant + ONE normal beta-globin gene; clinically normal (no anaemia requiring treatment in most); HPLC: HbA2 elevated (3.5–7.0% — hallmark of BTT); Hb mildly reduced (10–13 g/dL); MCV <75 fL (microcytosis); RBC count high; NO symptoms; CARRIER can pass mutation to children | BTI: hydroxyurea (increases HbF — reduces erythropoietic drive — reduces iron absorption); iron chelation (start at ferritin >800 µg/L even without transfusions); folic acid; splenectomy (if massive splenomegaly causing hypersplenism — reducing transfusion requirement); avoid iron supplements (iron already absorbed in excess); BTT (carrier/trait): no treatment required; health education: MUST screen partner before marriage if BTT — if partner also BTT (25% risk BTM child per pregnancy → offer prenatal diagnosis); avoid unnecessary iron supplementation (many Indian BTT carriers given iron supplements for mild anaemia — dangerous as iron NOT deficient — give HbA2 HPLC FIRST before iron in all microcytic anaemia) | BTT mismanagement India: largest single clinical error — microcytic hypochromic anaemia → empirical iron given without HPLC → BTT patient given unnecessary iron → iron overload over years; India: serum ferritin should be checked before iron supplementation in ALL microcytic anaemia; HPLC mandatory before iron therapy in recurrent iron-deficiency-non-responsive anaemia; BTT HbA2 >3.5%: do NOT give iron (unless co-existing iron deficiency confirmed by serum ferritin <30); BTI management: evolving — Luspatercept (activin receptor ligand trap — Reblozyl) now approved for NTDT: reduces transfusion burden 70% in BELIEVE trial; rarely available/affordable India (>₹5 lakh/injection) |
| Alpha-Thalassaemia in India | Alpha-globin gene deletions (chromosome 16 — 4 alpha-globin genes — 2 per chromosome); mild deletions: alpha+ thalassaemia (–/αα or -α/αα) — silent carrier/trait — mild microcytosis, no clinical disease; severe deletions: alpha0 thalassaemia — HbH disease (3 gene deletion — β4 tetramers form = HbH — Hb 7–10 g/dL — haemolytic anaemia, splenomegaly, jaundice); Hydrops fetalis (Hb Bart’s — 4 gene deletion — no alpha chains at all — lethal in utero or at birth — incompatible with life — cardiac failure + hydrops); alpha-thalassaemia prevalence India: moderate — Northeast India (Assam, Manipur, NE tribal groups — higher alpha-thal prevalence); Orissa sickle belt: alpha-thal + sickle trait common (alpha-thal actually AMELIORATES sickle cell disease — reduces intracellular HbS concentration) | Alpha-thal trait (1 or 2 deletions): no treatment; carrier counselling; HbH disease: folic acid; avoid oxidant drugs (same as G6PD — some drugs worsen HbH haemolysis); transfusion if Hb <7; splenectomy (if severe hypersplenism); Hydrops fetalis (Hb Bart’s): prevention ONLY — prenatal diagnosis on CVS/amniocentesis → termination of affected pregnancies; alpha-thal DNA testing: PCR-based gap-PCR (detects alpha-gene deletions); HPLC alone insufficient for alpha-thal diagnosis (HbA2 may be NORMAL or low in alpha-thal — unlike beta-thal where HbA2 is HIGH); molecular genetics needed | Alpha-thal diagnosis India: under-recognised — most microcytic anaemic patients in NE India with normal HbA2 and negative iron studies are likely alpha-thal carriers; molecular genetic testing at CMC Vellore, AIIMS, PGIMER; Hydrops fetalis: still births + maternal morbidity (toxaemia of pregnancy) from Hb Bart’s — prenatal diagnosis + counselling prevents; alpha-thal + sickle: in some tribals (Odisha), alpha-thal co-inherited with HbS → milder SCD — HU still beneficial but SCD course naturally less severe |
| Haematopoietic Stem Cell Transplant (HSCT) — Cure for Thalassaemia Major | HSCT (bone marrow transplant — BMT): only currently available cure for thalassaemia major; replaces patient’s defective stem cells with donor stem cells (ideally HLA-matched sibling) → donor produces normal haemoglobin lifelong; Lucarelli Classification (risk stratification for HSCT in thalassaemia): Class I (no hepatomegaly, no portal fibrosis, good chelation compliance): OS 93%, EFS 91% — excellent outcome; Class II: intermediate; Class III (hepatomegaly + portal fibrosis + poor chelation): EFS 79% — still excellent in experienced centres; Conditioning regimen: myeloablative — busulfan + cyclophosphamide + ± fludarabine; Matched sibling donor (MSD): preferred; matched unrelated donor (MUD): increasing success at specialised centres; haploidentical (half-matched — parent): newer option — expanding donor pool; age: best outcomes if transplant performed before 5–7 years (Class I/II) — before significant iron overload damage; in early >10 year olds: increased transplant complications but still curative benefit | HSCT outcomes India: 80–93% event-free survival in Class I/II patients at CMC Vellore, AIIMS Delhi, BGSLH Kolkata, TATA Memorial; cost: ₹15–30 lakh (government centre — AIIMS CMC subsidised) to ₹40–60 lakh (private); PMJAY: thalassaemia HSCT covered under PMJAY from 2020 (up to ₹5 lakh limit — often inadequate); fundraising through Thalassaemia Society of India; Aga Khan University Hospital (Pakistan) — used by some Indian thalassaemia patients pre-COVID at lower cost; eligibility: HLA-matched sibling required (only 25% of siblings are HLA-identical — limiting factor); without matched sibling: matched unrelated donor (bone marrow registry — DATRI India donor registry — 300,000+ registered unrelated donors); DATRI: Donors Associations and Tissue Recipients of India — India’s largest stem cell donor registry | India HSCT access: fewer than 500–700 thalassaemia HSCT performed per year (vs 10,000–15,000 new BTM births/year) — massive gap; primary barrier: cost (₹15–30 lakh at government); only 25% have HLA-matched sibling; inadequate donor registries; post-HSCT care: graft-versus-host disease (GVHD) prevention (cyclosporin, methotrexate); rejection monitoring (chimerism testing — proportion of donor vs host cells); if rejection: iron overload continues — restart chelation; long-term: regular transfusion-free life; normal endocrine function if transplanted early before iron damage |
| Gene Therapy — Emerging Cure | lentiGlobin (Zynteglo — bluebird bio): lentiviral vector delivers functional beta-globin gene into patient’s own stem cells (autologous — no donor needed); FDA/EMA approved 2022; Phase 3 HGB-207 trial: 91% of BTM patients became transfusion-independent (HbA-T87Q production); CRISPR-Cas9 gene editing: Casgevy (exa-cel — Vertex + CRISPR Therapeutics): reactivates foetal haemoglobin (BCL11A erythroid enhancer disruption → HbF persistently elevated → compensates for absent HbA); FDA approved December 2023 (first approved CRISPR medicine in world) for both SCD and BTM; price: $2.2 million USD (single treatment, lifelong cure) in USA — completely inaccessible to India currently; clinical trials India: AIIMS Delhi participating in gene therapy trials (Sangamo Therapeutics ST-400); CRISPR India: ICMR-NII collaborating with domestic CRISPR platform development (IISER Pune — CRISPR for haemoglobinopathies — 5–10 year horizon for affordable India version) | Current gene therapy: not available as standard care India (2026 — clinical trial phase only); prevention remains primary strategy for India: universal carrier screening + prenatal diagnosis prevents need for gene therapy lifelong; hydroxyurea: increases HbF enough in SCD/NTDT to provide significant benefit — partially mimics gene therapy HbF-induction mechanism at a fraction of the cost (₹300/month vs ₹18 crore for Casgevy); future India affordability: domestic CRISPR gene therapy may reach ₹50–100 lakh within 10 years if Indian manufacturers develop equivalent platform | India gene therapy status: no gene therapy approved or commercially available (2026); AIIMS HSCT + gene therapy research unit: clinical trials open for eligible BTM patients; hope horizon: by 2030–2035, if CRISPR costs fall dramatically (similar to how HIV antiretroviral went from $15,000/year to $100/year in India within 20 years via generic manufacturing), domestic gene therapy could be accessible; Thalassaemia Society India: tracking gene therapy trials globally; international collaboration: India researchers contributing to global trial design for resource-limited setting adaptation |
Frequently Asked Questions
How is thalassaemia prevented in India — and what should a carrier couple do?
Prevention of thalassaemia major births is the most cost-effective strategy for addressing India’s thalassaemia burden — far cheaper than treating each affected child for a lifetime. The science and the tools are available; what remains is political will, systems-level implementation, and community education: The cascade of prevention: Step 1 — Carrier screening: HPLC testing of all individuals before marriage or at first antenatal visit; HPLC identifies BTT (beta-thalassaemia trait): HbA2 >3.5%, microcytic hypochromic anaemia; carrier status is silent — no symptoms — only identifiable through HPLC; Step 2 — Partner testing: if one partner is BTT → test other partner immediately (same HPLC); if both BTT → 25% risk of BTM child per pregnancy + 50% risk of BTT carrier child; Step 3 — Genetic counselling: for BTT × BTT couples: explain Mendelian inheritance; risk per pregnancy: 25% BTM (severely affected — needs lifelong transfusion), 50% BTT (carrier — healthy), 25% HbAA (completely normal); Step 4 — Prenatal diagnosis (PND): options: Chorionic villus sampling (CVS): 10–12 weeks gestation (safest for PND — enough foetal DNA +results before 14 weeks → allows early termination if required); amniocentesis: 15–18 weeks; foetal blood sampling (cordocentesis — 18+ weeks — rarely used for thalassaemia); testing method: DNA-based molecular genetics — beta-globin gene mutation analysis (Sanger sequencing or next-generation sequencing panel — NGS); identifies specific mutations in foetal sample → predicts HbAA/BTT/BTM genotype; Step 5 — Termination of affected pregnancies: if BTM foetus → couple informed and offered termination; Medical Termination of Pregnancy Act India 2021 (amended): allows termination up to 24 weeks for substantial foetal abnormality including genetic disease; HPLC alone of foetal blood: no longer used (inadequate in early foetal blood — DNA required). India systems gaps in thalassaemia prevention: Carrier screening: not universal — many couples discover BTT × BTT status only after birth of first BTM child; premarital HPLC: mandatory only in Gujarat (pilot), Rajasthan (partially) — no national mandate; antenatal HPLC: patchy — available at medical college cities but not at PHC/urban CHC level; MTP Act 2021 — 24-week provision: critical win for thalassaemia PND (CVS at 11 weeks + result by 13 weeks + termination at 14 weeks = well within 24-week limit); awareness gap: many rural communities do not know what thalassaemia trait is; some communities (e.g., Sindhi, Punjabi communities) have excellent awareness due to Thalassaemia Society activism; cost of PND: CVS + DNA analysis: ₹8,000–25,000 at government/private hospitals — free at AIIMS Delhi, CMC Vellore, PGIMER Chandigarh for eligible patients; premarital screening campaign India: launched by Ministry of Health 2023 — awareness drive targeting high-prevalence communities.
What is iron overload in thalassaemia — and how is iron chelation monitored?
Iron overload is the most dangerous complication of thalassaemia major — a slow, insidious accumulation of iron in vital organs (heart, liver, endocrine glands) from each blood transfusion that, without chelation, leads to organ failure and premature death, typically in the 2nd–3rd decade of life: How iron overload develops in thalassaemia: Each unit of packed red blood cells (250 mL — approximately 200 mL of red cells) contains approximately 200–225 mg of elemental iron; the human body has NO physiological mechanism to excrete excess iron — only absorbs and stores it; a thalassaemia major patient receiving 20 units/year accumulates approximately 4,000–4,500 mg of iron per year; over 10 years: 40,000–45,000 mg of excess iron (body contains only 3,000–4,000 mg normally); additionally: ineffective erythropoiesis in thalassaemia → HEPCIDIN suppression → increased dietary iron absorption by gut (even without transfusion) → NTDT patients also develop iron overload. Organs affected by iron overload and their consequences: Heart (most critical — leading cause of death): iron deposits in cardiomyocytes → fibrosis → restrictive/dilated cardiomyopathy → heart failure + arrhythmias; cardiac MRI T2* (milliseconds): the best tool: T2* >20ms = normal iron; T2* 10–20ms = mild cardiac iron; T2* <10ms = severe cardiac iron → urgent intensify chelation + specialist cardiologist referral; T2* <6ms = very severe → highest mortality risk; Liver: iron deposits → hepatic fibrosis → cirrhosis; LIC (liver iron concentration) by liver MRI T2* or R2 (FerriScan): normal <3 mg/g dry weight; therapeutic target <7; severe >15; liver biopsy: historically used, now largely replaced by MRI; Endocrine glands: pancreas (diabetes mellitus — type 3c — iron-induced beta-cell failure), pituitary (GnRH/FSH/LH → delayed puberty, infertility, hypogonadism), thyroid, adrenals, parathyroid; monitoring endocrine function: annual GTT (glucose tolerance test), LH/FSH/testosterone/estradiol, thyroid function. Iron chelation types, monitoring, and targets: Deferasirox (DFX — oral tablet, once daily): 20–40 mg/kg/day; side effects: Fanconi syndrome (proximal renal tubular acidosis), elevated creatinine (dose-dependent), GI upset, skin rash; monitor creatinine + urine protein monthly initially; reduce dose if creatinine rises >33%; Deferoxamine (DFO — SC overnight infusion): gold standard for severe cardiac iron (T2* <10ms) + DFP combination; ototoxicity (audiogram annually) + retinal toxicity (ophthalmology annual); urine colour: red-brown (“vin rosé” colour) from ferrioxamine excretion — reassuring sign that chelation is working; Deferiprone (DFP oral TDS): strongest cardiac iron chelation; side effects: agranulocytosis (1%) — MANDATORY weekly CBC on DFP; arthropathy (joint pain); GI; Monitor: serum ferritin monthly (target <1000 µg/L); T2* cardiac MRI annually (target >20ms); LIC by MRI annually; endocrine panel annually; ophthalmology + audiology annually; echocardiogram annually.
What to Read Next
- Sickle Cell Disease — Compare SCD vs Thalassaemia: Both Beta-Globin Disorders; HU Works in Both; SCD = VOC + ACS; Thal = Transfusion + Iron Overload
- Anaemia — Critical Distinction: Iron Deficiency (Low Ferritin + Low HbA2) vs BTT (Normal Ferritin + High HbA2) vs Both Combined; HPLC First
- Pregnancy — Thalassaemia Major + Pregnancy: Extreme High-Risk; Chelation Hold (DFX Teratogenic); Transfuse to Hb ≥10; Cardiac Iron Pre-Conception Assess
- Diabetes — Thalassaemia Diabetes (Type 3c): Iron Overload β-Cell Failure; Insulin Dependent; Not Type 2; SGLT2i Contraindicated (Iron Overload Risk); HbA1c Unreliable in Thal
- Liver Cirrhosis — Thalassaemia Iron-Induced Cirrhosis: Distinct from Viral Hepatitis; MRI LIC Instead of Biopsy; Intensify Chelation + Monitor AFP for HCC
A Sindhi couple in Pune — both doctors — are planning their first pregnancy. Routine HPLC: both BTT. HbA2: 5.1% and 4.7%. Genetic counsellor: “Every pregnancy carries a 25% chance of a child who will need blood transfusions every 3 weeks for life.” They opt for prenatal diagnosis via CVS at 11 weeks. DNA analysis: BTM (compound heterozygous — IVS 1-5 G→C + Codon 8/9 +G). They discuss with counsellors for two weeks. They terminate at 13 weeks. Their next pregnancy is screened: BTT (carrier — healthy child). Their daughter is born healthy. She is a thalassaemia carrier — she will know, and she will screen her future partner. Each screened couple breaks the chain. India’s 10,000 BTM births per year → potential zero — if every pregnant woman gets HPLC.
About This Guide: Written by the StudyHub Health Editorial Team (studyhub.net.in) based on Thalassaemia International Federation (TIF) Guidelines 2021, India National Thalassaemia Control Programme 2022, ICMR Task Force Haemoglobinopathies India 2022, and ASH Thalassaemia Guidelines 2020. Last updated: March 2026.
🧬 Planning Pregnancy? Get HPLC Thalassaemia Carrier Test Now: HPLC (haemoglobin analysis) is the only reliable test for thalassaemia trait. Available at most hospital labs (₹400–800). If your HPLC shows HbA2 >3.5% — you are a thalassaemia carrier. Your partner must be tested BEFORE pregnancy. If BOTH are carriers: prenatal diagnosis via CVS at 11–12 weeks can identify affected foetuses. Free HPLC + genetic counselling at AIIMS, CMC Vellore, PGIMER — contact your nearest government medical college thalassaemia clinic.
⚠️ Microcytic Anaemia? Get HPLC BEFORE Taking Iron: If you have microcytic hypochromic anaemia (low MCV on CBC) and your doctor wants to prescribe iron tablets — ask for HPLC first. If you have thalassaemia trait (HbA2 >3.5%), your iron stores are NORMAL or HIGH — taking iron supplements is dangerous. Do NOT take iron for thalassaemia trait anaemia.
⚕️ Medical Disclaimer: This article provides general educational information about thalassaemia. Diagnosis (HPLC + DNA), transfusion management, chelation initiation/monitoring, prenatal diagnosis, and HSCT eligibility assessment require qualified haematologist evaluation. Chelation drugs (deferasirox, deferoxamine, deferiprone) have significant side effects requiring specialist monitoring.
